The Making of a Cleanser

The purpose of a cleanser and how it works is rather simple. But formulating one is not.
It should be effective enough to remove all unwanted oil residues from skin, but not too effective that it removes too much of our skin’s natural oils.
Given the importance of surfactants in cleansers, it comes as no surprise that they greatly affect how cleansers perform.
Types of surfactants
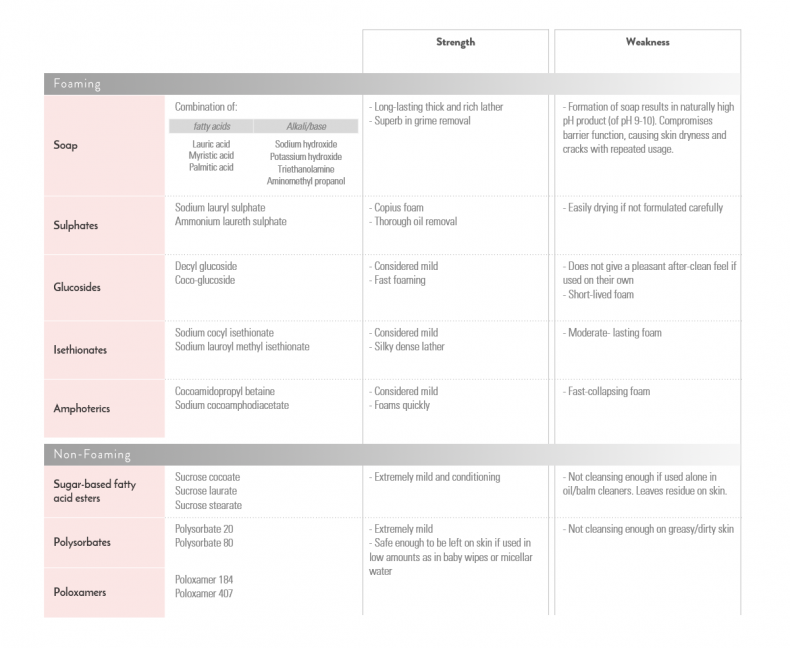
There are many different types of surfactants, from very high foaming to none at all. Generally, the higher the degree of foaming, the stronger its cleansing power.
Robust foaming ones clean excellently but might strip skin oils excessively, while extremely mild surfactants may not be enough to adequately remove everything. These are the reasons why sulphate/soap-containing products can feel drying and balm/oil/cream cleansers leave a residue on skin respectively.
The image below lists some common ones used in cosmetics. Each type has its own set of strengths and weaknesses, making formulating a good cleanser rather complex.
Other considerations
Although surfactants are the key functional ingredients in cleansers, other factors also play a part in the overall formula.
Skin protective emollients and humectants counteract the stripping effects of surfactants, allowing a satisfying foamy wash without overstripping. Having a pH close to that of healthy skin (4.5-5.5) is also important in maintaining a strong and intact skin barrier.
The perfect cleanser
You can see now that the perfect cleanser combines the right amounts of surfactants with skin protective ingredients . The key is thus to find one with the best combination for your skin.
If you want to skip the trouble of searching for it, let us custom formulate your cleanser for you. This way, you can be sure that it is going to be perfect for your skin.
I hope this has given you some insight into the formulation behind a seemingly uncomplicated product!